|
Simply take each element and multiple it by the number of times the bracketed structure occurs. We don't have brackets implemented (yet), so you will need to unpack any bracketed expressions. It solves for total mass of a molecular formula (average molecular weight).įrom there we break the formula for Ammonium nitrite into parts -Ī Nitrogen atom, a Hydrogen atom, a Oxygen atom, etc. 
The molar mass calculator - it will show the count of atoms, the atomic weight of each element, and the molecular weight for the molecule. We present the results in a table at the bottom of Then weĬompare each atom against a table of the standard atomic weights for that element. 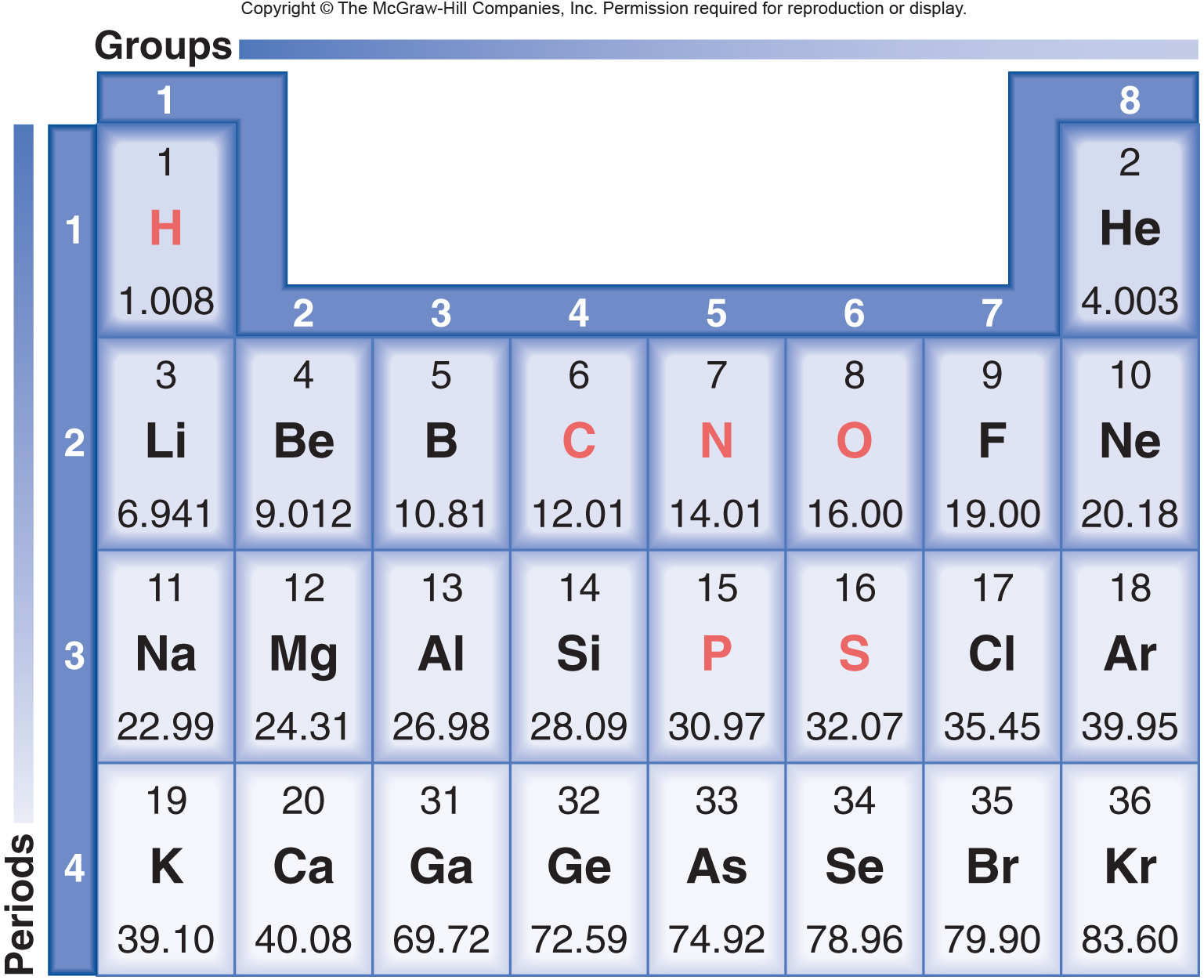
We take the formula you provide (NaCl - common table salt - in our default example) and unpack it into the component elements. Other terms: atomic mass of Ammonium nitrite, molar mass of Ammonium nitrite, molecular mass, Use the mole ratio and empirical formula to understand the limits of the reactants. Percent yield calculator which can help you apply this to actual experiments. This project started with as a molar mass calculator for chemical reactions. Note that the calculator assumes a pure substance - if you'reĪware of dilution or impurities, make appropriate adjustments for the molarity of a given substance. The calculator takes the elemental composition of the compound and weighs the elements to get an The molar mass of a chemical compound based on the compound's empirical formula. Need to know the atomic mass of a Ammonium nitrite molecule? Our molar mass calculator uses the periodic table and the chemical formula to solve for 
Solving for the atomic mass of Ammonium nitrite (NH4NO2)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
